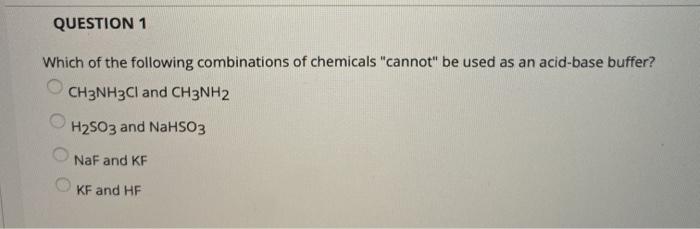
SOLVED: Rank the following salts in order of increasing pH a) NaNO2, CsCl, CH3NH3Cl b) Ca(ClO4), K2S, NH4NO3

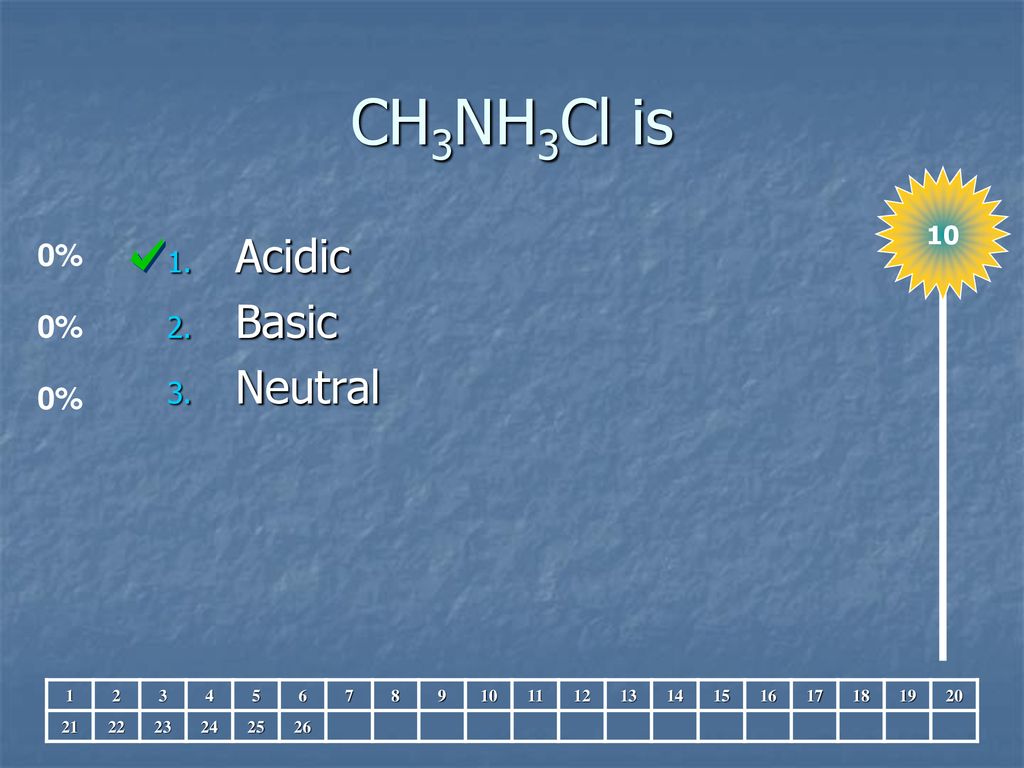
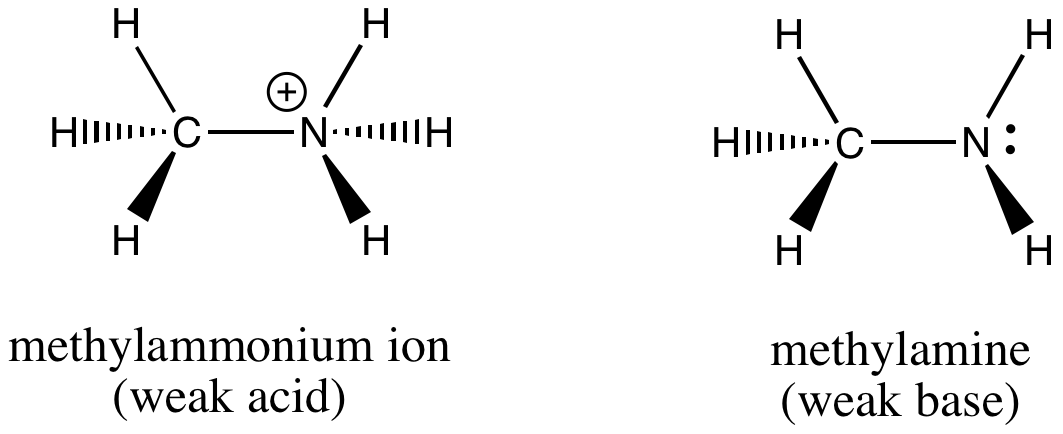
SOLVED: Methylamine, CH3NH2, and methylammonium chloride, CH3NH3Cl write the identity of the two species in each set (strong acid/strong base/weak acid/weak base/salt/other) and explain reason. Identify if anything in common betweenthe two

Solved) - Is the solution of KCI acidic, basic or neutral?. Is the solution... (1 Answer) | Transtutors

OneClass: please write an EQUILIBRIUM reaction that shows CH3NH3Cl asacidic- Write a chemical equatio...

CH3NH3Cl-Assisted One-Step Solution Growth of CH3NH3PbI3: Structure, Charge-Carrier Dynamics, and Photovoltaic Properties of Perovskite Solar Cells | The Journal of Physical Chemistry C