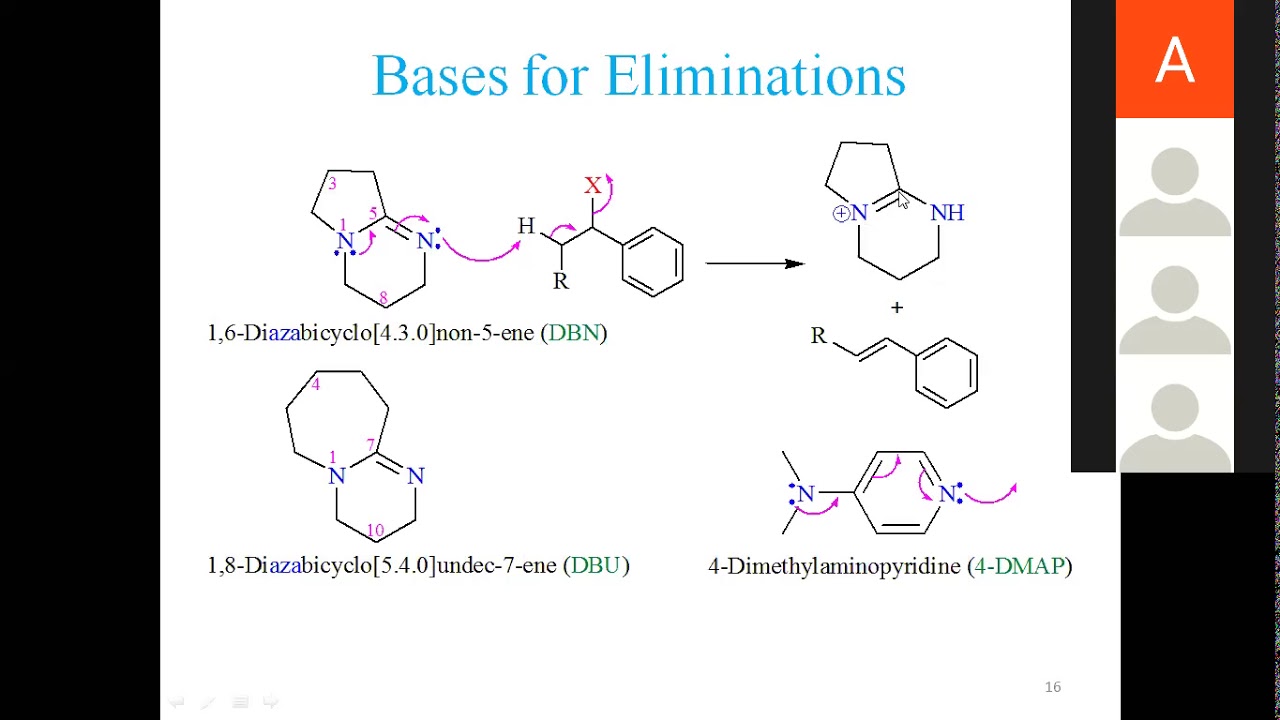
![Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/da071cf8-eee0-4e2f-8e22-5b6f6a0f91e7/adsc201601279-fig-5004-m.jpg)
Controlled Reactivity of 1,8‐Diazabicyclo[5.4.0]undec‐7‐ene (DBU) in the Selective Synthesis of 1‐(Bromoethynyl)arenes - Krishna Moodapelly - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library

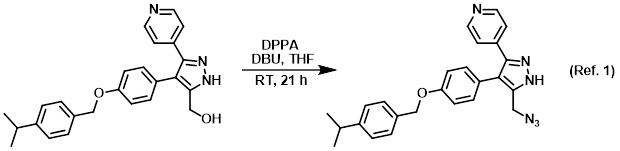
SciELO - Brasil - Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry and DFT Studies of Catalyst Role Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry
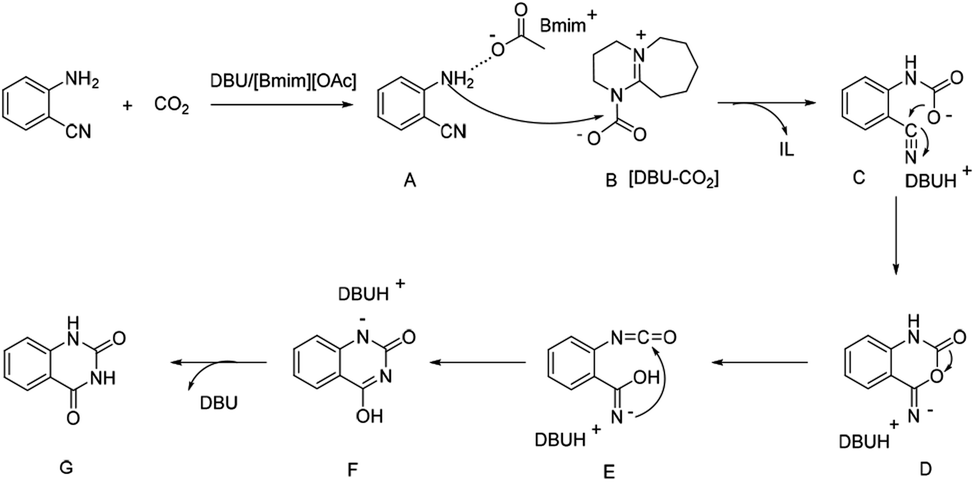
DBU coupled ionic liquid-catalyzed efficient synthesis of quinazolinones from CO 2 and 2-aminobenzonitriles under mild conditions - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00194E
![Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) | The Journal of Organic Chemistry Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.7b01530/asset/images/large/jo-2017-01530d_0002.jpeg)
Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) | The Journal of Organic Chemistry
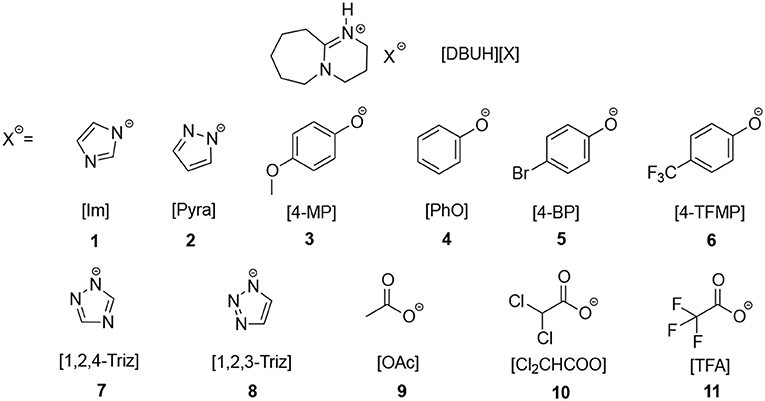
Frontiers | CO2 Absorption by DBU-Based Protic Ionic Liquids: Basicity of Anion Dictates the Absorption Capacity and Mechanism
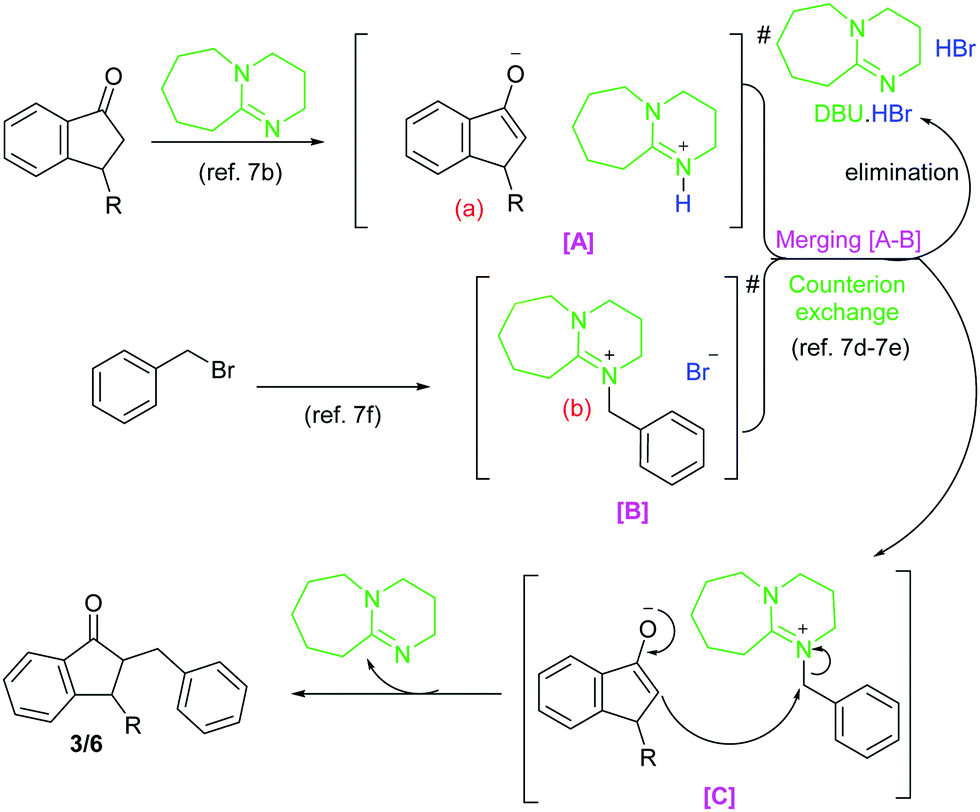
An efficient merging of DBU/enolate and DBU/benzyl bromide organocycles for the synthesis of alpha benzylated 1-indanone derivatives - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D2NJ00783E

N-Bromoimide/DBU Combination as a New Strategy for Intermolecular Allylic Amination | Organic Letters
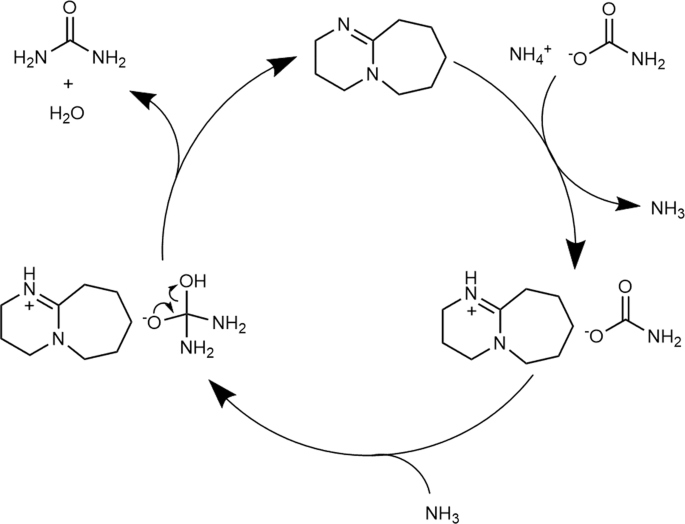
Organic bases catalyze the synthesis of urea from ammonium salts derived from recovered environmental ammonia | Scientific Reports
![Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences](https://royalsocietypublishing.org/cms/asset/0ea92727-dfe5-4dad-b4fd-c68f2d8d3d5e/rspa20190238f03.jpg)
Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
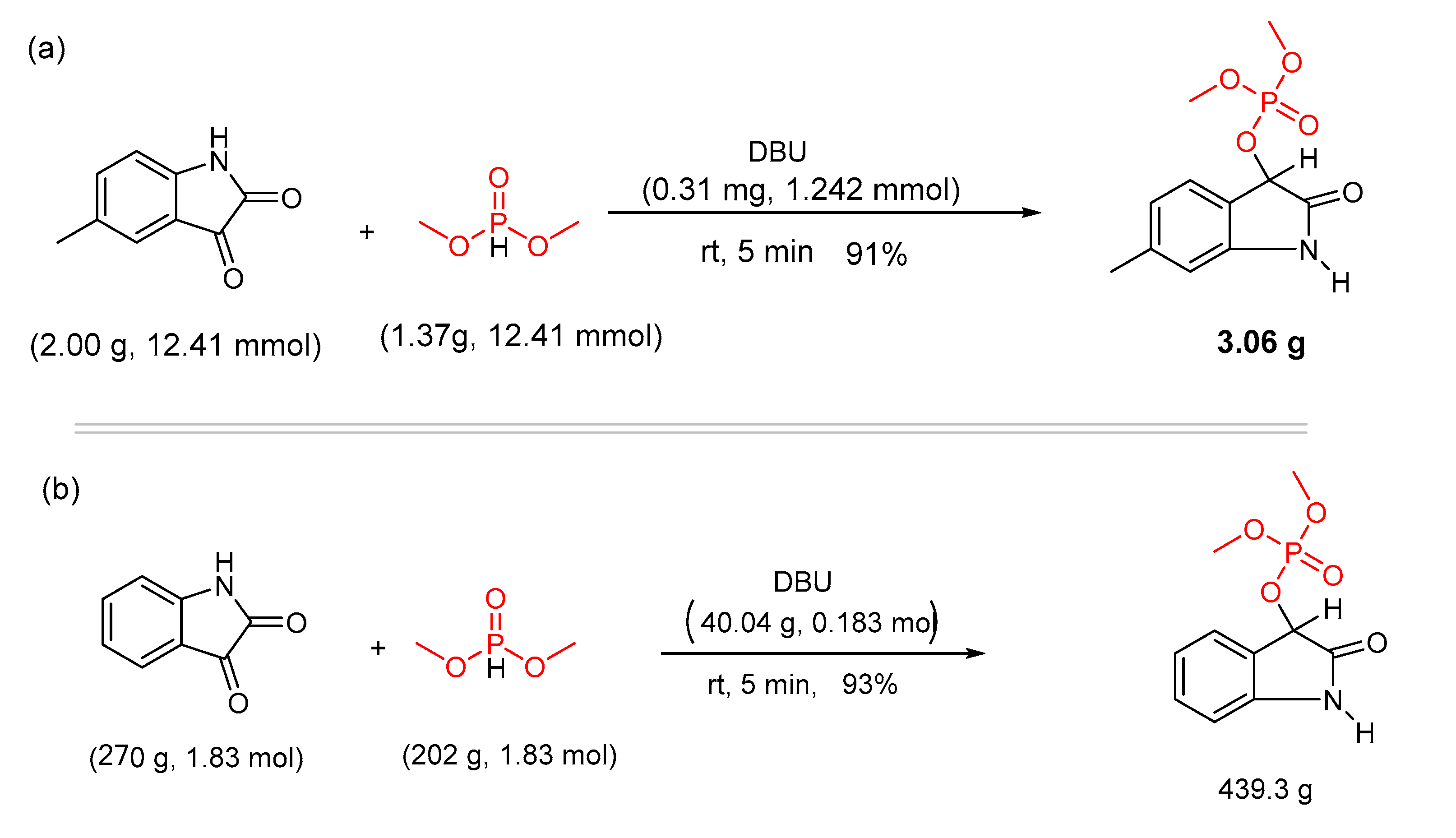
Catalysts | Free Full-Text | DBU Catalyzed Phospho-Aldol-Brook Rearrangement for Rapid Preparation of α-Phosphates Amide in Solvent-Free Conditions

NBS/DBU-Promoted One-Pot Three-Component Cycloaddition of Malonic Acid Derivatives, Nitrosoarenes, and Alkenes: Synthesis of Isoxazolidines | The Journal of Organic Chemistry
![The Dual Role of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the Synthesis of Terminal Aryl- and Styryl-Acetylenes via Umpolung Reactivity | Organic Letters The Dual Role of 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) in the Synthesis of Terminal Aryl- and Styryl-Acetylenes via Umpolung Reactivity | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.5b02398/asset/images/large/ol-2015-02398j_0006.jpeg)