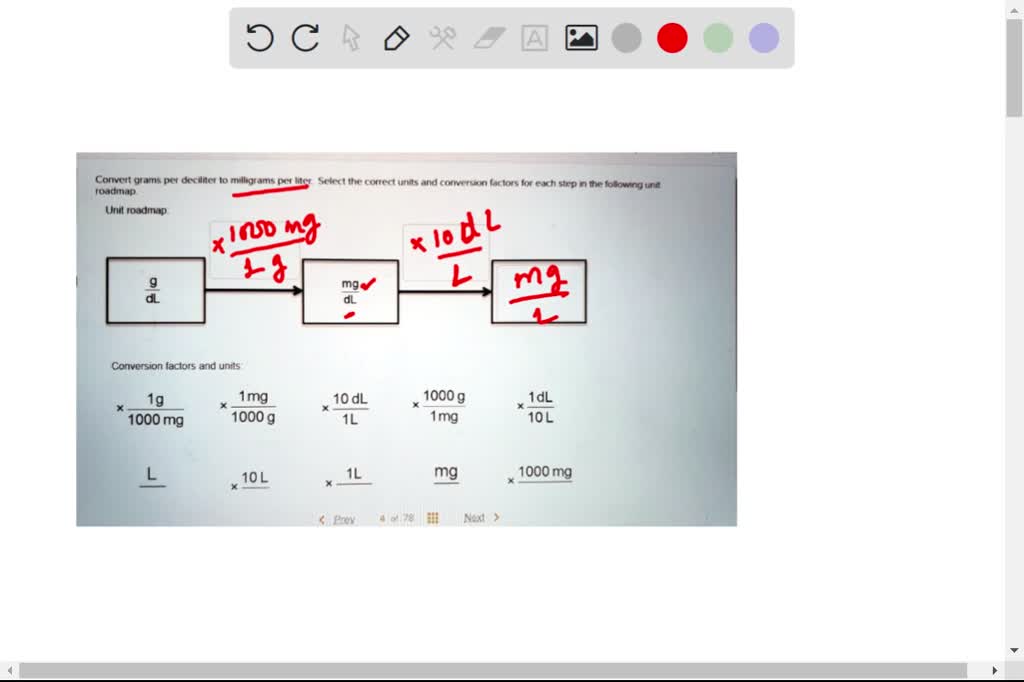
SOLVED: Convert grams per deciliter millligrams per litcr Select the correct units and corversion factors for cach step in the following unit roadmap Unit roadmap mg Conversion factors arid units 1g 1mg

Comparison of specific growth rate (μ, liters per hour), volumetric... | Download Scientific Diagram

SOLVED: points) Initially 15 grams of salt are dissolved into 10 liters of water: Brine with concentration of salt 3 grams per liter is added at a rate of 4 liters per

SOLVED:Calculate the solubility of each of the following compounds in moles per liter and grams per liter. (Ignore any acid-base properties.) a. A g3 P O4, Ks p=1.8 ×10^-18 b. CaCO3, Ksp=8.7 ×

Solutions A solution is formed when a substance is dissolved in a liquid. The concentration of the solution may be expressed as – grams per Litre g L ppt download

Concentration. Concentration Particles per volume Can be in grams per litre but chemists usually express concentration in moles per litre This are related. - ppt download