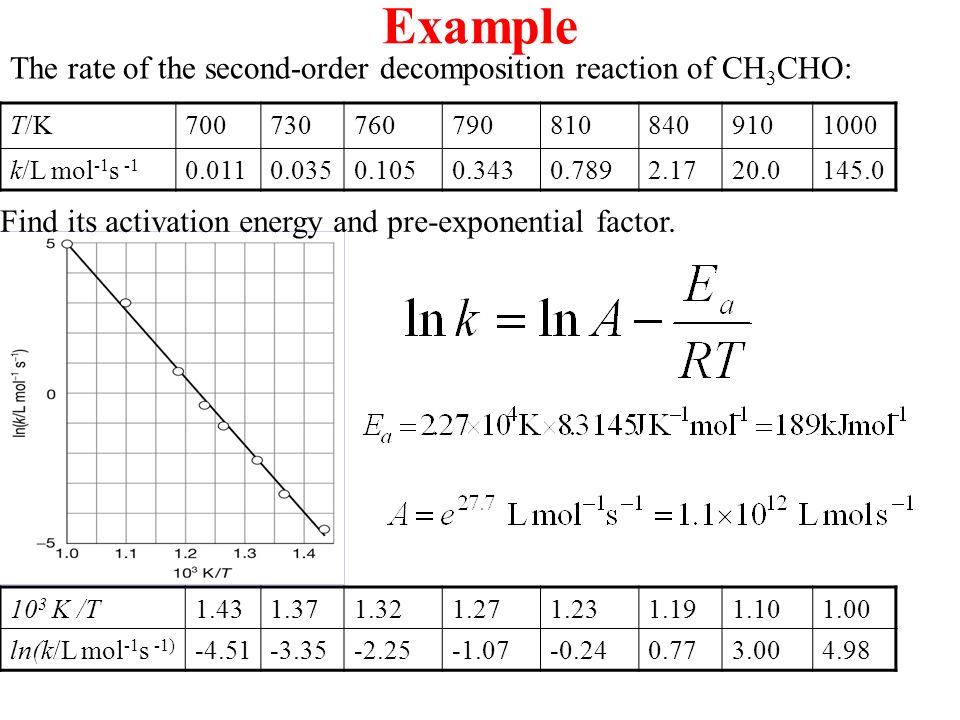
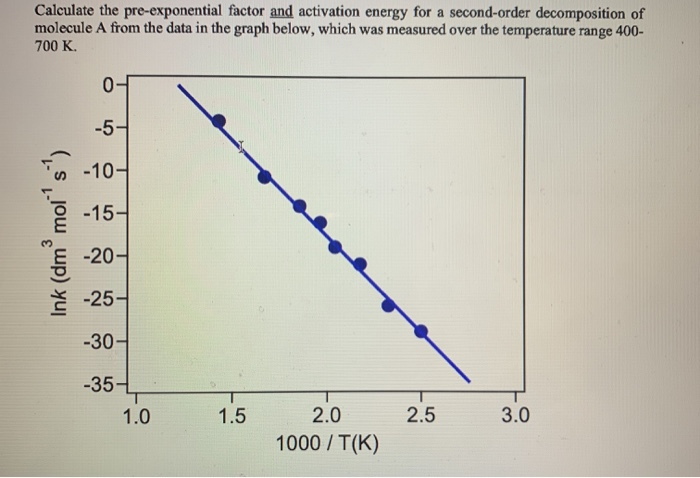
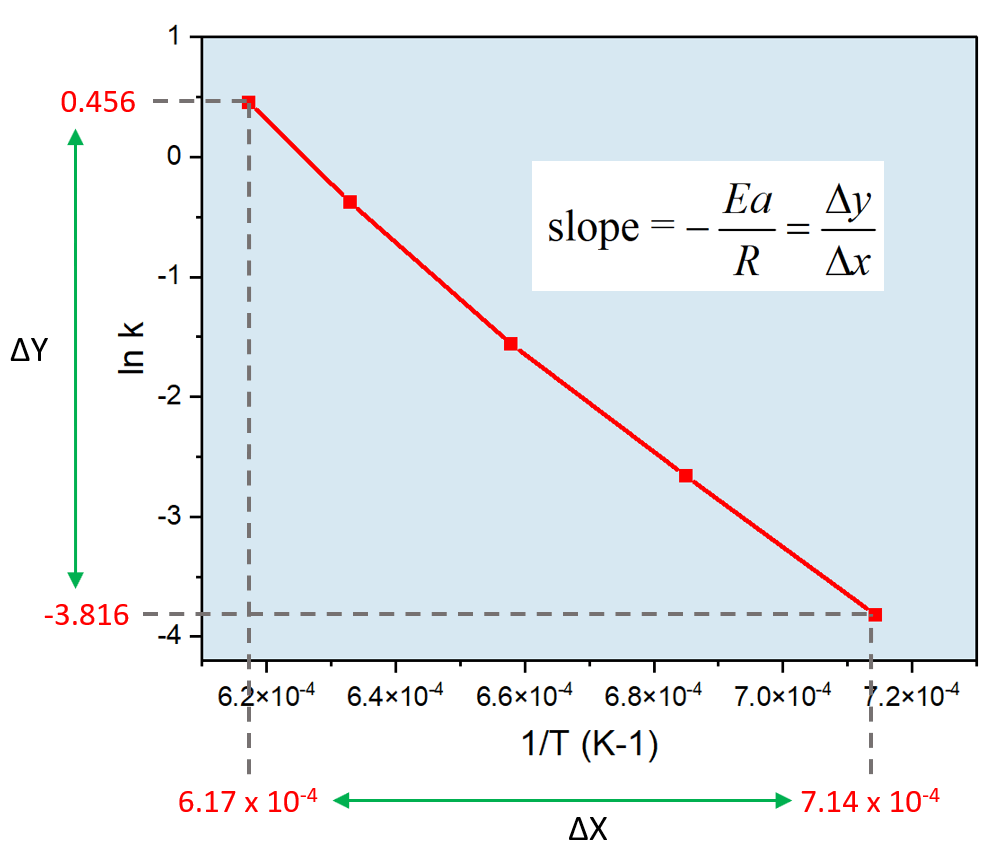
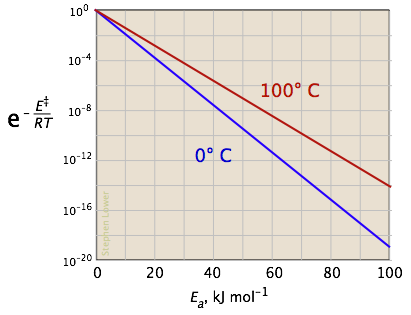
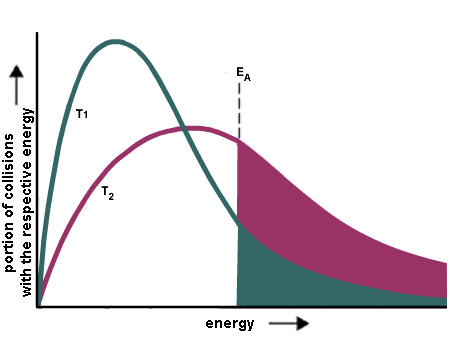
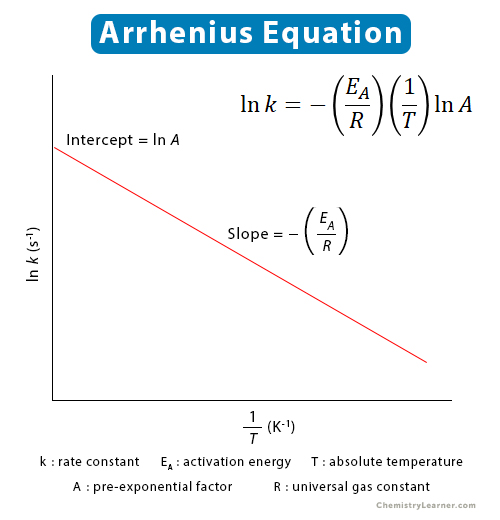
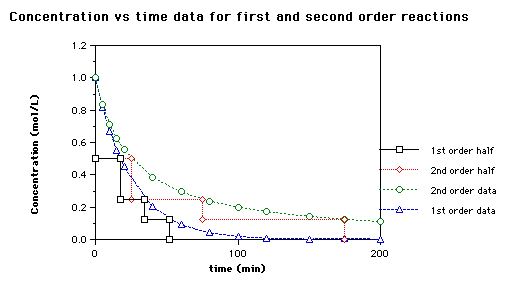
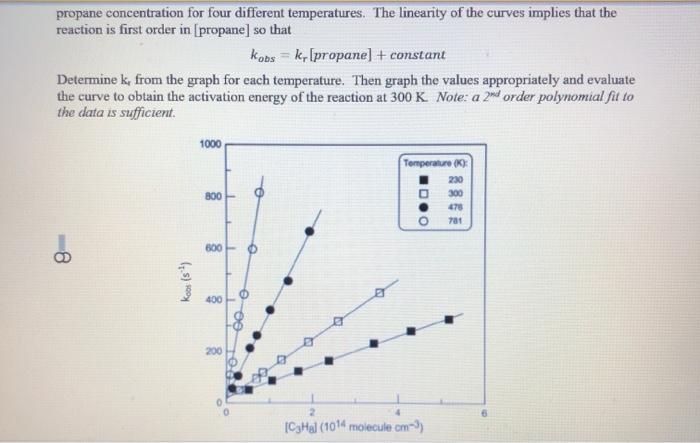
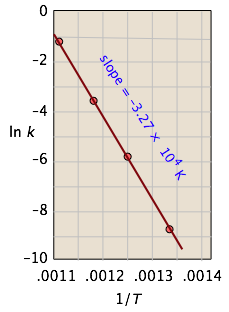
The rate constant for the decomposition of hydrocarbons is 2.418 × 10^-5s^-1 at 546 K . If the energy of activation is 179.9 kJ/mol , what will be the value of pre - exponential factor?
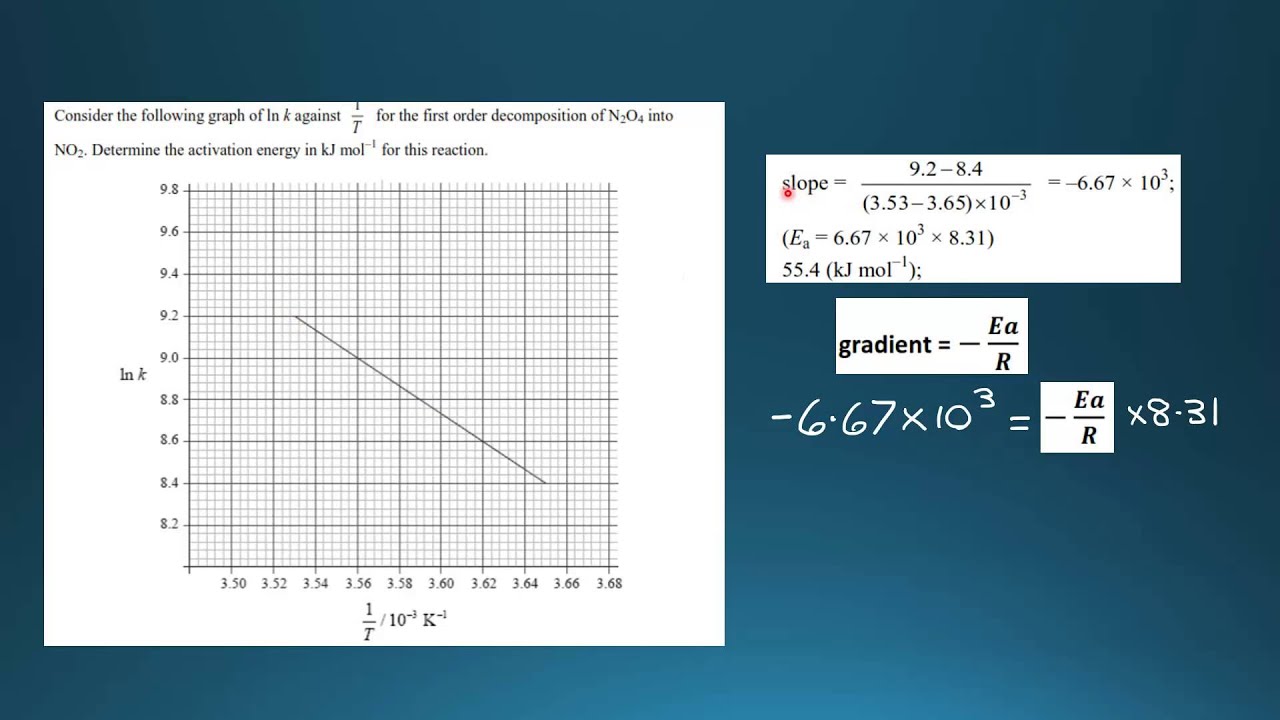
16.3.2 Determine activation energy (Ea) values from the Arrhenius equation by a graphical method. - YouTube

Collision Theory and activation energy|Pre exponential factor Arrhenius equation|Rank Booster - YouTube
Chemical Kinetics Goal: Our goal in this activity is to explore some applications of chemical kinetics. We begin by thinking c

Molecules | Free Full-Text | Advanced Isoconversional Kinetic Analysis for the Elucidation of Complex Reaction Mechanisms: A New Method for the Identification of Rate-Limiting Steps
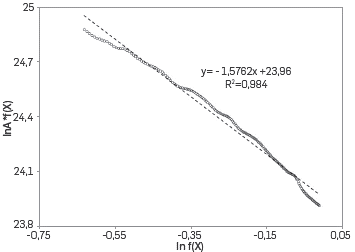
METHODOLOGY FOR CALCULATING THE PRE-EXPONENTIAL FACTOR USING THE ISOCONVERSIONAL PRINCIPLE FOR THE NUMERICAL SIMULATION OF THE AIR INJECTION PROCESS