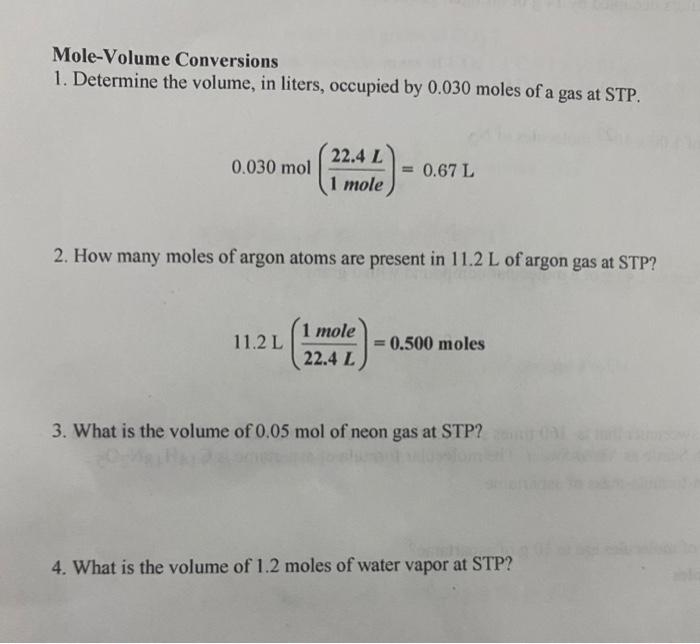The number of water molecules in 1 litre of water is - Sarthaks eConnect | Largest Online Education Community

An equilibrium mixture in a vessel of capacity 100 litre contains 1 mol N2 , 2 mol O2 and 3 mol NO . No. of moles of O2 to be added so
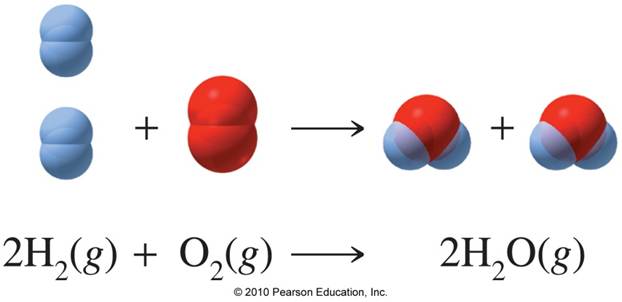
How many liters of water can be made from 55 grams of oxygen gas and an excess of hydrogen at a pressure of 12.4 atm and a temperature of 85C? | Socratic
calculate the no of moles present in 1 litre of water if the dw c alculate the no of moles present in 1 l of water if the density of water is

SOLVED: Mole-Volume Conversions Determine the volume in liters occupied by 0.030 moles of a gas at STP. How many moles of argon atoms are present in 11.2 L of argon gas at




:max_bytes(150000):strip_icc()/GettyImages-692027135-419fe3ddc26e4415b356380582c4e5b2.jpg)