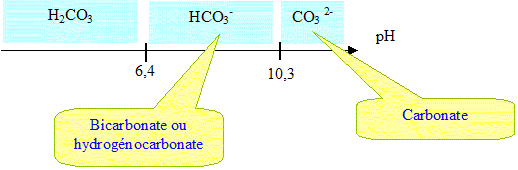
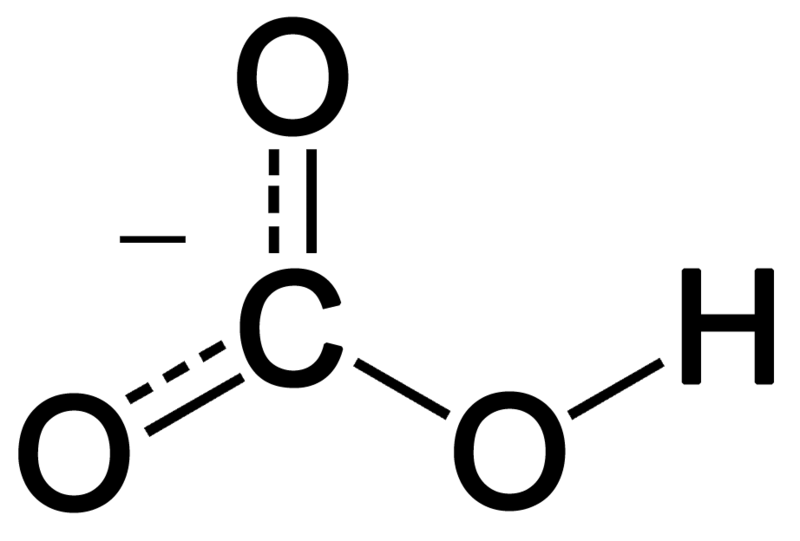
30 1. L'équation de dissolution dans l'eau de l'hydrogénocarbonate s'écrit : NaHCO (s) → Na+ (aq) + HCO 2. Pour dim
I. Quelles sont les réactions qui échangent des protons ? Exercice A1 Exercice A2 : Couples acides-bases conjugués. Exercice

Write the separate reactions of cinnamic acid with sodium bicarbonate and sodium hydroxide. What is the gas that is evolved when cinnamic acid reacts with sodium bicarbonate? | Homework.Study.com

Total Synthesis of 6-Deoxyerythronolide B via C–C Bond-Forming Transfer Hydrogenation | Journal of the American Chemical Society

Write the separate reactions of cinnamic acid with sodium bicarbonate and sodium hydroxide. What is the gas that is evolved when cinnamic acid reacts with sodium bicarbonate? | Homework.Study.com
41 1. L'hydrogénocarbonate de sodium NaHCO (s) est insoluble dans l'huile mais soluble dans l'eau. Lorsque la goutte de v

Vidéo de question : Déterminer les produits formés à partir de la réaction entre le carbonate de sodium et l'acide chlorhydrique | Nagwa