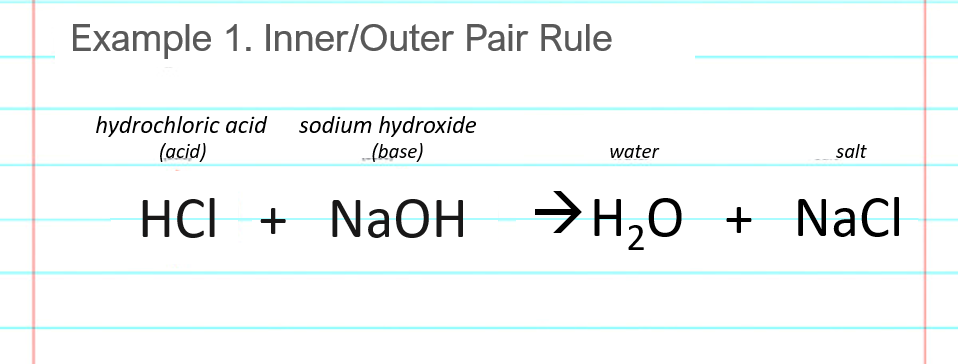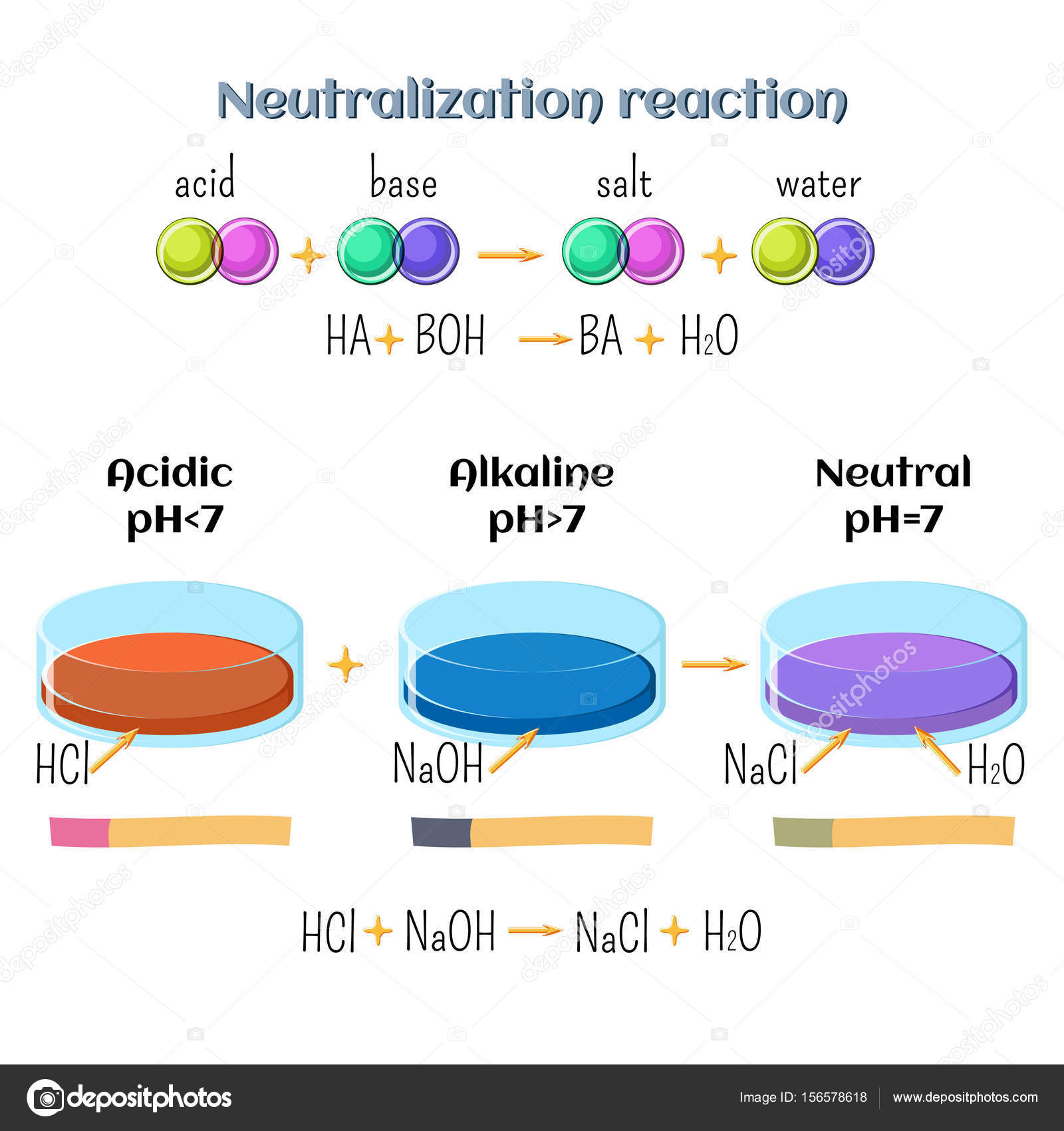
Acid-base, neutralization reaction of hydrochloric acid and sodium hydroxide. Types of chemical reactions, part 6 of 7. Stock Vector Image by ©inkoly #156578618

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa

Acid – base reaction. chemical reaction neutralization the acid and base properties, producing a salt and water. used to determine pH. Bronsted – Lowry theory. molecules of HCl, NaOH, H2O, and NaCl,
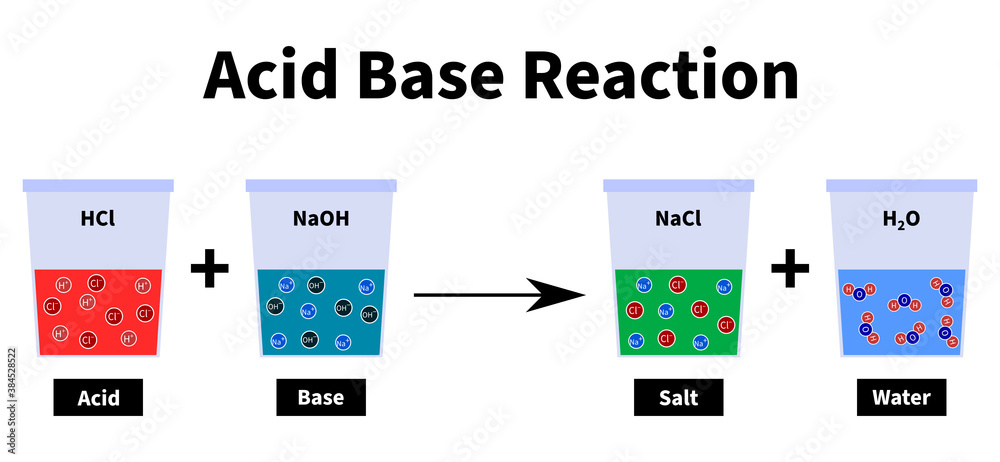
Acid base reaction: salt water hydrogen chloride sodium hydroxide sodium chloride water, neuatralization, chemical reaction Stock Illustration | Adobe Stock

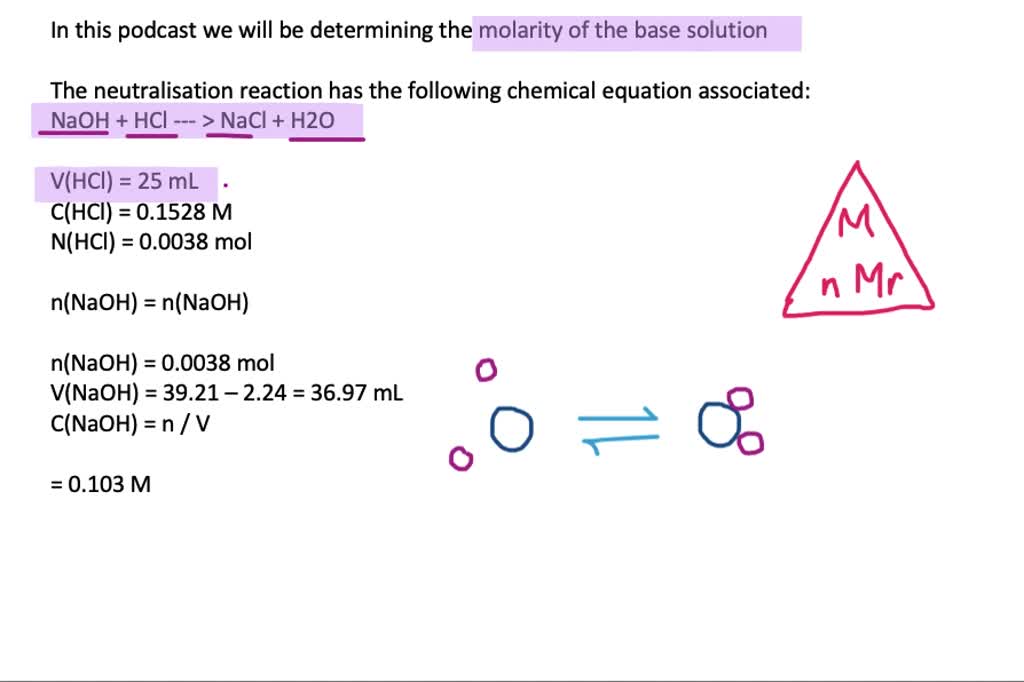
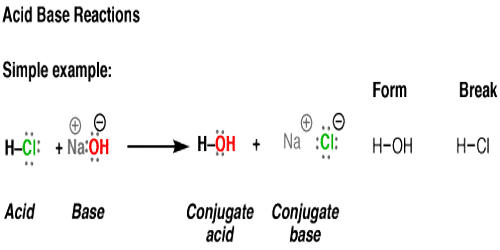
Write the neutralization reaction between Hydrochloric acid HCI and sodium hydroxide NaOH, and write the equation for this process.

Selective focus of sodium hydroxide base and sulfuric acid solution in brown glass and plastic bottle inside a chemistry laboratory. White background Stock Photo - Alamy
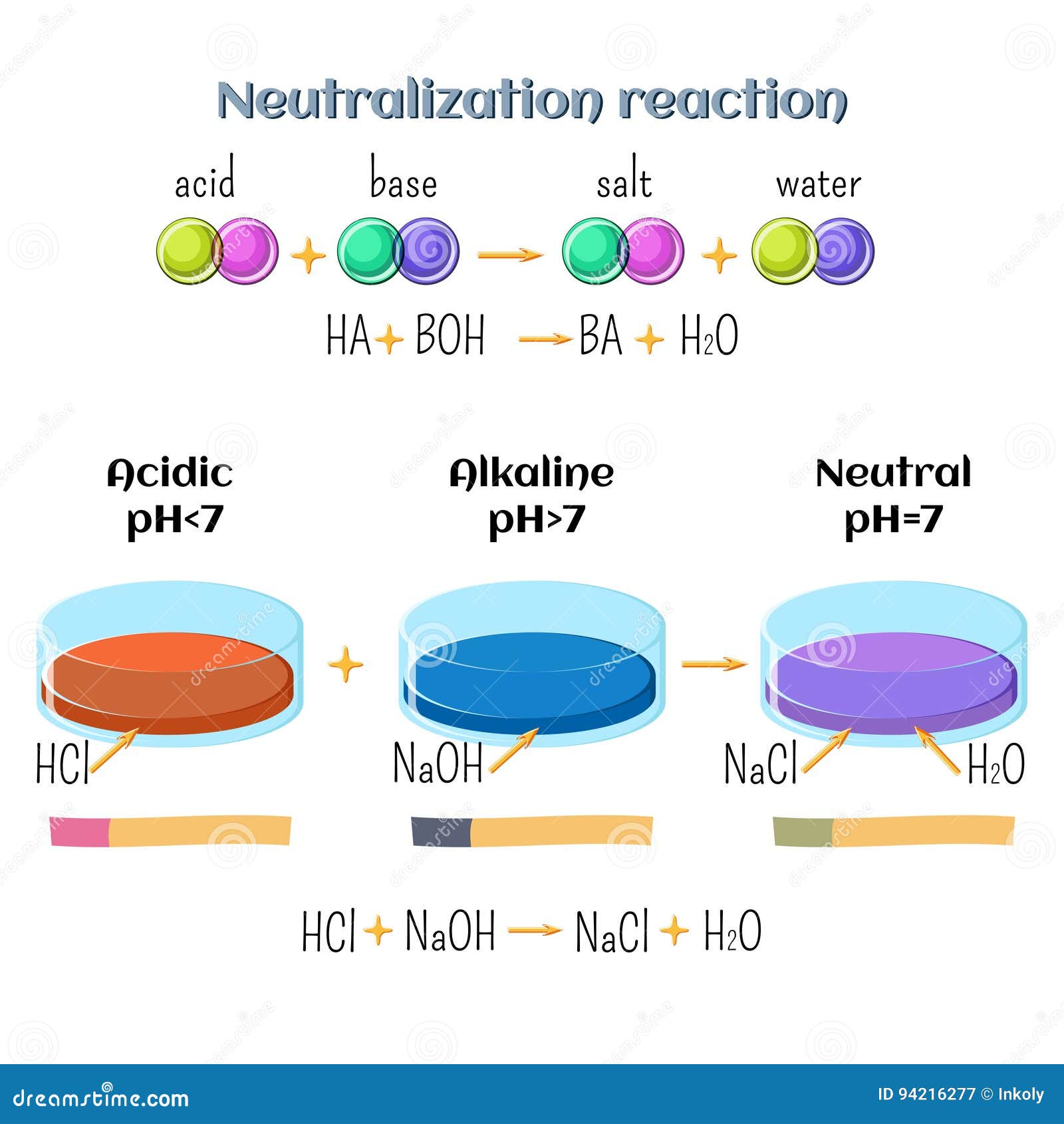
Acid-base, Neutralization Reaction of Hydrochloric Acid and Sodium Hydroxide. Types of Chemical Reactions, Part 6 of 7 Stock Vector - Illustration of acid, atom: 94216277